
With the necessary approvals in place, Dana Farber/Boston Children’s collaborated with other gene therapy centers to open an international clinical trial in 2010. In 2008, buoyed by the discovery of safer and more effective vectors, Boston Children’s Hospital researchers requested permission from the government to revive this potentially life-saving treatment for SCID-X1. The tragedy cast a pall on - and temporarily stalled - gene therapy research, leading to stricter regulations. Although almost all of the boys had improvement of their immune system, one-quarter of the patients developed treatment-related leukemia. Paris and London investigators conducted parallel trials using a retrovirus-derived vector (or vehicle). The development of gene therapy strategies to treat SCID-X1 began in Europe during the late 90s. The history of stem cell gene therapy is forever linked to SCID-X1 or X-linked SCID, the first inherited condition in which gene therapy in hematopoietic stem cells was performed successfully. The evolution of X-linked SCID gene therapy Learn more about the Gene Therapy Program. He’s just a very lovable boy, and we are grateful parents. He loves cars - anything with wheels - and animals. He’s a perfect 18-month old boy, always with a smile on his face. Today, his immune system is normal, and he doesn’t take any medications. And as of January 2019, Paul is no longer on isolation. We knew we’d be in the hospital for at least a month, and we also knew Paul would experience some type of pain from the chemotherapy, but eventually he was well enough to be discharged on May 20. 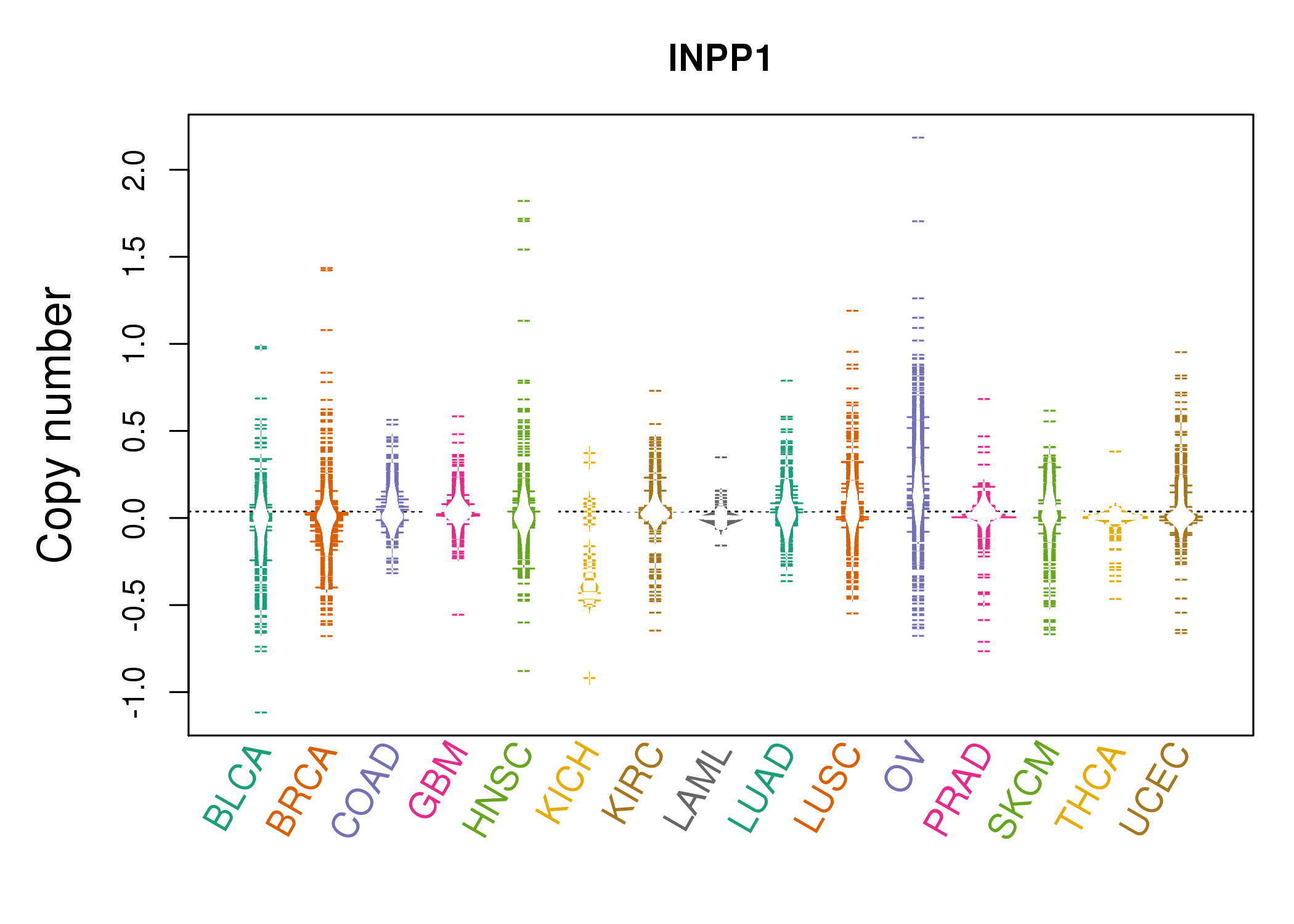
This is called his “life day” - day zero. Then, at last, on April 20, Paul received his genetically-modified stem cells. 
The following day, it was time for his body to rest. On April 17 and 18, Paul was given chemotherapy. 
Pai comforted us, and the wait was worth it. We were nervous, anxious, and scared, but Dr. It seemed like forever for this day to arrive. We waited a month for them to reprogram the cells and checked back in for the long haul on April 16, 2018. Marrow to reprogram them with the purpose of repairing the faulty gene that They extracted his stem cells from the bone The gene therapy trial beginsĪt 4 months old, we checked in overnight at Boston Children’s Hospital, so theyĬould harvest Paul’s bone marrow. Between the air filter and seemingly endless cleaning, miraculously, we kept Paul from getting sick until he was ready for his treatment. We brought him home to a sterile environment with a high-powered air filter and a year’s supply of Lysol wipes, donated to us by a charity called Cameron’s Crusaders. Kevin, Paul, and LaDreama with their care teamĪlong with Paul’s doctors, we came up with a birth plan to have Paul at UMASS Memorial in Worcester, Massachusetts. Trial, but the word “trial” scared us, so at first, we didn’t focus on that We also were made aware of a gene therapy clinical Pai, we talked mainly about the standard treatmentįor this disease - a bone marrow transplant - and were given a detailed description Pai at Dana Farber/Boston Children’s CancerĪnd Blood Disorders Center. Shortly after finding out, the two of us, accompaniedīy Paul’s grandmother, would have our first appointment with Dr. Weeks into the pregnancy we learned our baby would be born with SCID. A difficult decision: Bone marrow transplant vs. The typical treatment is a bone marrow transplant, but we opted to enroll Paul in a gene therapy clinical trial, and his progress has been amazing. “bubble-boy disease,” because children with SCID have to be kept isolated to His condition isĬombined immunodeficiency (SCID). Spirit, yet without an immune system - meaning that the simplest infection wasĭifficult to fight and a common cold was life threatening. Just 18 months, our son Paul Gallagher has been through more than what most
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
